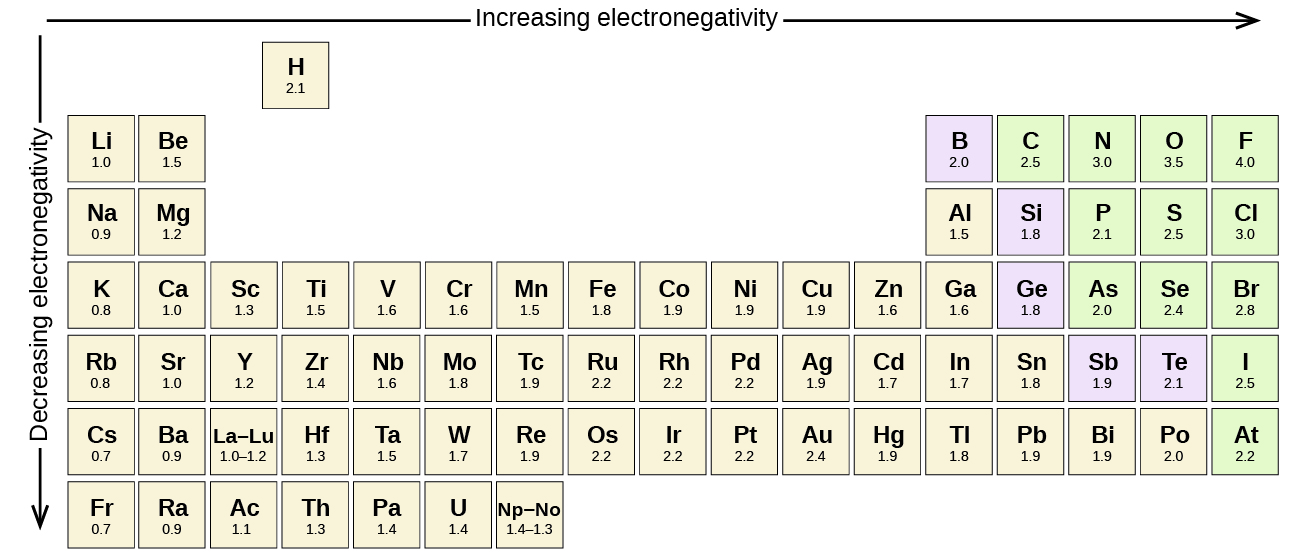
Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. 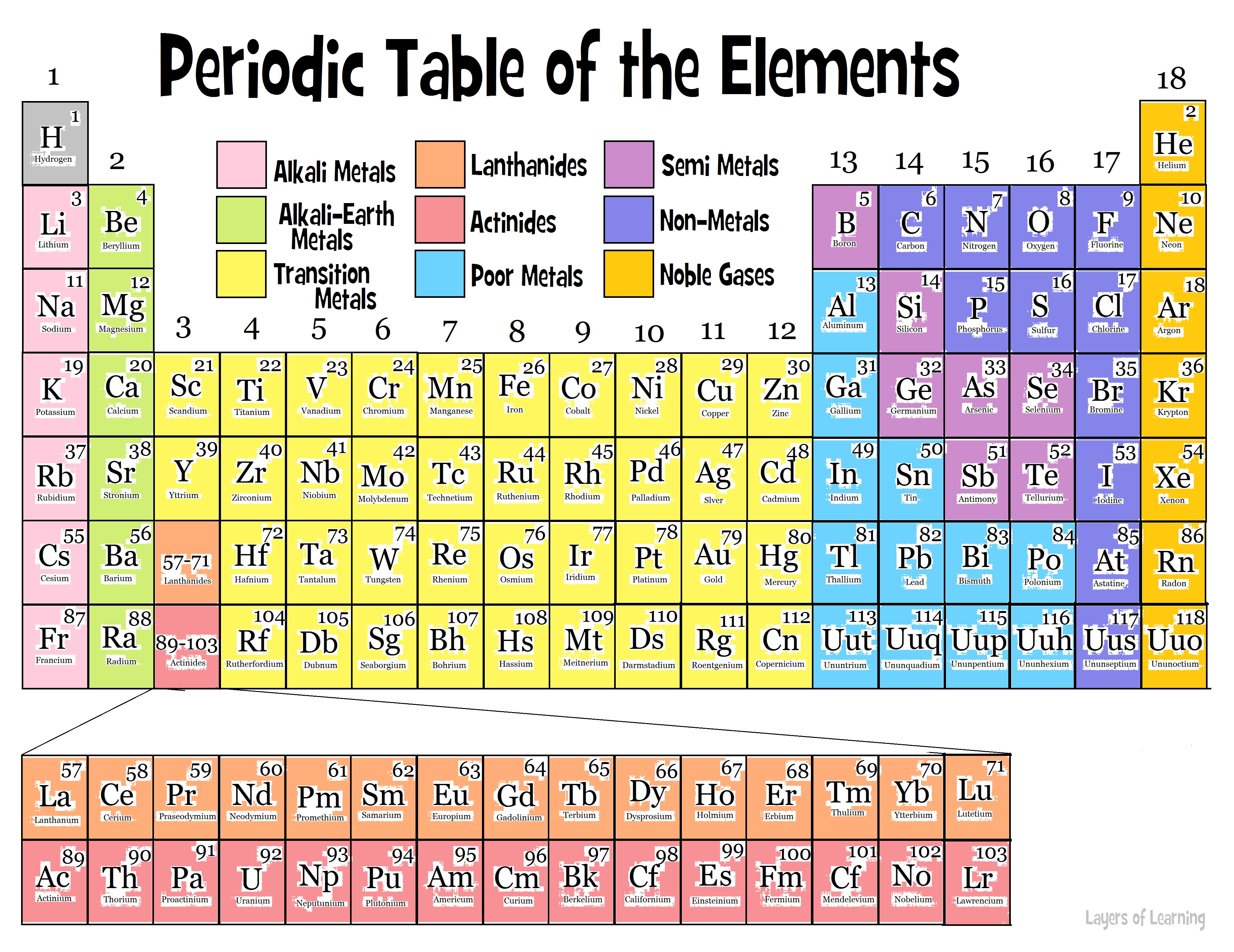
Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. 1984, 61(2), 136-136 ( ).The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital. Loening, “Recommended Format for the Periodic Table of the Elements,” J. Powell, “Confusion in the periodic table of the elements,” J. Horace Grove Deming, General Chemistry J.Fluck, “New notations in the periodic table,” Pure and Applied Chemistry 1988, 60(3), 431-436 ( ). Thus, it is safe to say that this nomenclature is now outdated and not worth discussing. The IUPAC Commission on the Nomenclature of Inorganic Chemistry (CNIC) proposed the designations of groups by Arabic numerals in 1984 (Ref.4), which was approved by the Nomenclature Commission of the American Chemical Society (ACS) and rest is history. Moreover, more than 10% of the articles it was nearly impossible, from the wording of the text, to recognize which elements were being discussed without further information (Ref.3). An investigation of the application of the subgroup designations A and B in all articles, which appeared between 19 and covered by Chemical Abstracts, revealed a completely arbitrary use of the designation. The designations A and B have been extensively and rather arbitrarily used in the meantime in textbooks and in publications. This version of the periodic table was distributed for many years by the Sargent-Welch Scientific Company, Skokie, Illinois, USA. The group was later switched to the right side and usually labeled as Group VlllA. The Noble Gas Group was originally attached by Ueming to the left side of the periodic table. The iron, cobalt, and nickel groups were designated neither A nor B. The numeration was chosen so that the characteristic oxides of the B groups would correspond to those of the A groups. Deming used the long periodic table in his textbook General Chemistry (See following diagram with Roman numerals only Ref.2), which appeared in the USA for the first time in 1923, and designated the first two and the last five Main Groups with the notation "A", and the intervening Transition Groups with the notation "B". 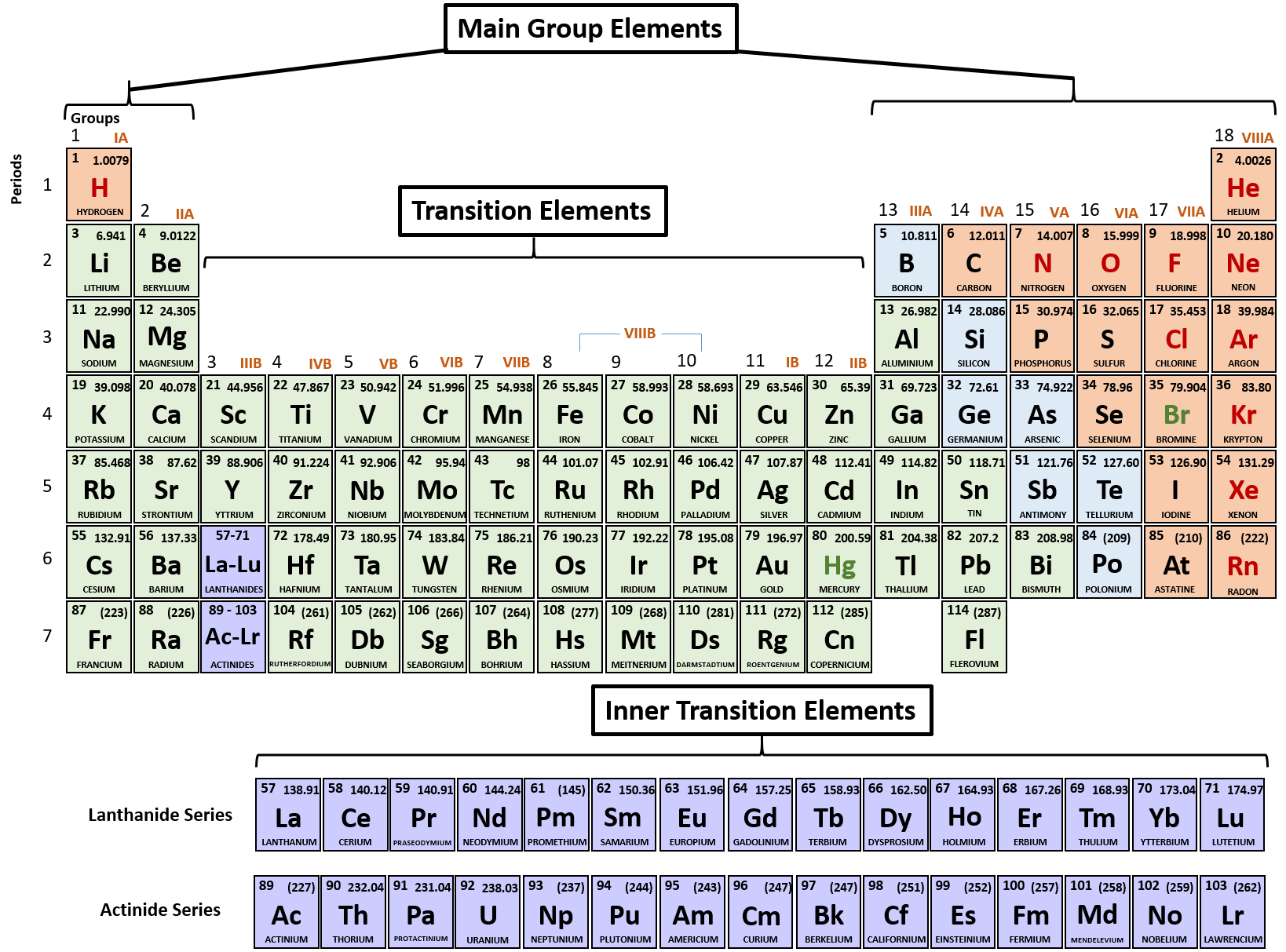
The IUPAC recommended Periodic Table is given below (ignore the Roman numerals, which I put in there to explain):Īccording to Ref.1, the confusion of Periodic Table of Elements has been addressed as follows: I believe I-VIII Roman numeral nomenclature belong to CAS system while 1-18 Arabic numeral groups are recommended by IUPAC (Ref.1). Even though your question get downvoted, I think it is an legitimate question for people to understand the trend.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
